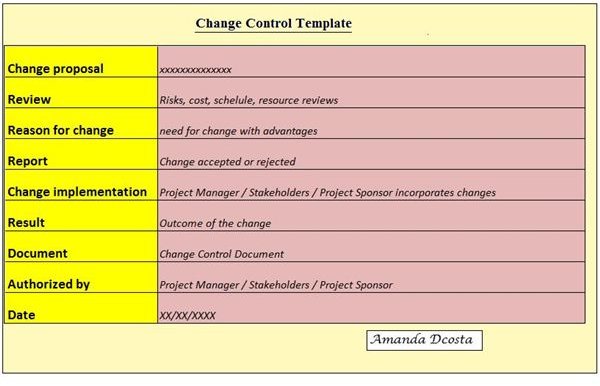
A change control document is part of a change process that takes place in a project. There is a specific format that takes a project through a change in any given phase which is officially documented.
What is a change control document?
Projects are susceptible to changes right from the initiation and planning phases. While changes can be made with ease during these early phases, no changes can be made after the scope statement of work , project charter and work breakdown structure have been finalized. If there are any changes that have to be made due to unforeseen reasons on account of unidentified risks, or external or internal factors, changes have to be approved by all or majority of the stakeholders along with the project sponsor. There is a process for changes in a project and these changes are recorded in the change control document as per the change control plan . There is a specific process for change.
[caption id="" align=“aligncenter” width=“600”]
Procedure for Documentation
A change control document has to be a systematic and sequential entry of data supporting change in a project. It goes through an initial process of change request, through the process of change approval or denial and finally with change implementation, if it is approved. Noted below are a few basic steps of the change control documentation process. A sample change control documentation index is also available for download from Bright Hub’s media gallery . All entries are chronologically entered with the date. A name is assigned to this document, e.g., Change proposal for Phase 5 of the East Wing; Bay View Towers. Change proposal: This is the initial step where the project manager or any of the stakeholders request a change. It is only through the process of a proposal or a request can a change be considered. Reason for change: When the change is proposed, it should always be supported with a reason. Are the costs for a particular phase or the project running beyond expected limits? Does the project need additional manpower and why? Is the government withdrawing support for a particular project thereby forcing change in the project conditions? Review: A change that is proposed has to be reviewed. Various parameters such as cost, time, resource, manpower and technology along with their risks are reviewed and evaluated. The essence for this review is to analyze whether the proposed change is worth the effort or will it eventually be a drawback to the project. Report: Once a review is conducted, a report of the review is documented. All the findings, input, feedback and risk assessments are recorded. Change implementation proposal: How will the change be made? What are the steps to be taken to effect positive change in a project or project phase? Which departments will be responsible for implementing the change and who will be involved? How will the project manager assign duties for change control tasks? Such are the points that encompass change implementation. Change approval / denial: Once the changes have been reviewed and analyzed by the project manager, stake holders and project sponsor, it is either approved or denied based on numerical data mainly. If it is approved, it follows the following steps, or it will conclude with a ‘Change Proposal Denied’ status. Authorization: The project sponsor is the person responsible for authorizing change. It is the project sponsor who provides the additional funds that might be needed on account of this change and hence has to authorize this change. Implementation and result: What is the outcome of the change? There has to be a clear connection between the change proposal and the end result. Stakeholder support: All stakeholders who have been involved directly with the change process have to sign the change control document in support of the change. Likewise, the project manager will finally sign the document.
Conclusion
When the changes are chronologically entered, it is very easy to follow-up on information and be able to also gather information sequentially. A change control document not only provides the information pertaining to the change but also gives clarity and transparency to the process of change that has taken place with accountability.
Credit and Reference
Image Credit: Author, Amanda Dcosta Reference: Document and Change Control - FDA.gov